If you’ve found yourself around the world of cancer treatment recently, you’ve likely heard about the promise of immunotherapy. While conventional treatment has long been a combination of chemotherapy and radiation, re-equipping your immune system to fight tumor cells is biotech’s hottest strategy within the past few years, and holds promise looking forward. Immunotherapy offers doctors and their patients a much more specific, programmable, and localized treatment plan. Under the umbrella of immunotherapy exists BiTe’s, or Bi-Specific T-Cell Engagers. Below we’ll explain what that acronym means within your body, who it potentially helps, and what companies lead the way with BiTe Therapy. As always, I try to present the topic with nuance and thus point out why every Biotech company isn’t dropping their current pipeline to patent their own BiTe, as well as general shortcomings of the therapy.
Classroom Time
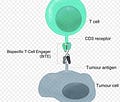
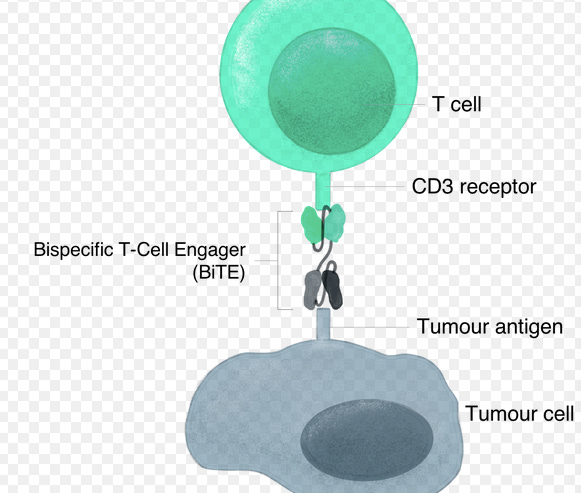
The current pandemic has likely exposed most of the general public to the idea and importance of antibodies. Take monoclonal antibodies for COVID patients, a treatment option to supply your body with the right type and quantity of antibody to enhance your depleted immune system. Moving forward, the most important characteristic of monoclonal antibodies in this context to remember is their antigen (think germ) binding site is largely unique to only one type of antigen. With BiTe’s the same antibody can bind to two different types of antigens at the same time. The clinical application of this technology in the cancer sphere is that BiTe’s can “grab” a tumor cell, while simultaneously “grabbing”/engaging a T-Cell. Together, these steps allow T-Cell to be selectively recruited to tumor sites and kill these same tumor cells. The bi-functional nature of these manufactured antibodies allows scientists to take a step beyond simple antibody cocktails to recruit antibodies to tumor cells or re-designing CAR T-Cells. Instead, T-Cell recruitment via BiTe’s allows selective activation of the immune system’s most potent defender, T-Cells, at the tumor site.
Promising Disease Targets
The first BiTe to be approved for clinical trials was Blinatumomab, licensed by Amgen for the treatment of B-Cell derived malignancies and Acute Lymphoblastic Leukemia (ALL). At time of writing, there are currently 15 clinical trials listed on ClinicalTrials.gov that involve Blinatumomab. For a deep dive on Blinatumomab you can read more here.
Much of the momentum surrounding BiTe therapy is driven through treatment of blood-derived tumors. For instance, Roche’s BiTe Mosunetuzumab had extremely positive Phase I/II results in pre-treated populations with Non-Hodgkin’s Lymphoma (NHL). For instance, mosunetuzumab “induced a complete and durable response lasting at least 18 months in patients who had already received two therapies”. NHL and blood cancers as a whole represent a sub-set of cancers that are quite resistant to conventional treatment modalities. As such, Mosunetuzumab is on an early track to be the first approved BiTe for NHL. Smarter and more seasoned minds than I are notably optimistic about Roche’s star.


A Dose of Reality
While positive results appear to exist for BiTe’s in clinical trials, there are a variety of disadvantages to address that more properly characterize the way in which this antibody fits into the treatment scene.
#1 Cytokine Release Syndrome
One of the most common clinical adverse events seen with BiTe treatment is Cytokine Release Syndrome (CRS). CRS is a inflammatory response occurring throughout that body, in this case in reaction to antibodies perceived as foreign. CRS can be fatal, and its management in clinic has become increasingly important for physicians involved in clinical trials. Given the complexity of bi-specific antibodies, they carry a much higher risk of CRS than conventional monoclonal antibodies. The mechanism by which CRS comes about is incompletely characterized in current literature. It is presently understood that the BiTe binds to one antigen, say a tumor cell, yet the other binding site is engaged by “bystander immune cells” and other off target cells that can lead to an overwhelming cytokine cascade. Downstream effects of this cascade can lead to both chronic and acute conditions.
#2 Bi-Specific vs Tri-Specific
While not inherently a weakness, the complexity of the immune system does not lend itself well to cookie-cutter solutions. For instance, the problem of CRS alone points out the limitations of bi-specificity. When the specificity of one binding site is not 100% accurate, we risk the possibility of activating non-target immune cells leading to disastrous cascades. From a pipeline development perspective, multi-functional antibodies offer another option for Biotech companies. For instance, why offer bi-specificity when you can manufacture tri-specific antibodies, turning appropriate arms on and off in specific disease settings. As such, a proper characterization of BiTe’s is that their potential efficacy in the clinic will lend itself to the creation of vastly more programmable and specific antibodies.
Future-Oriented Thoughts
It is easy to get caught up with the latest theoretical ways to tackle cancer. A main driving factor in this pigeonhole is the detrimental effects of shotgun therapies like radiation, and in a similar fashion chemo. While the media hype around novel science characterizes new therapies often as shotgun solutions to every cancer with incredible specificity, quick glances at clinical trial data bring almost all these drugs back to Earth. The manner in which Mosunetuzumab preformed in a specific ALL trial could reflect BiTe’s roadmap to patients around the globe. Instead of throwing antibodies at a variety of cancer types, perhaps the behavior and profile of blood cancers and NHL specifically lends itself well to T-cell engagement compared with general profiles of other cancers.
The trend toward individualized medicine doesn’t singularly have to be approached from the patient side. The use of personal medical tools and diagnostics helps immensely, and perhaps drug development could benefit from a more cell-individualized approach.

This is excellent work! Thank you for taking the time to research and write this. Extremely informative sir!